Chronic Pelvic Pain & Endometriosis
- Understand background, pathogenesis & physiological impact of endometriosis
- Understand the critical need for timely diagnosis and effective intervention
- Know where to address to
Endometriosis Defined
Endometriosis is an estrogen-dependent disease frequently resulting in substantial morbidity, severe chronic or recurrent pelvic pain, multiple surgeries, and impaired fertility.
Clinically Endometriosis is defined as presence of endometrial-like tissue (Endometrium lining like cells) found outside uterus/uterine cavity, resulting in sustained inflammatory reaction. Primarily the pelvic peritoneum, ovaries, and and the lowest abdominal cavity (rectovaginal septum, Pouch of Douglas) are affected.
Affecting 6%–10% of women of reproductive age, the stigmata of endometriosis include chronic pelvic pain in menstrual bleeding (dysmenorrhea), chronic pain while intercourse (dyspareunia), irregular uterine bleeding, and/or infertility.
The prevalence of this condition in women experiencing pain, infertility, or both is as high as 35%–50% Yet endometriosis is often under- or non-diagnosed and associated with a 6 to 7- year mean latency from onset of symptoms to definitive diagnosis
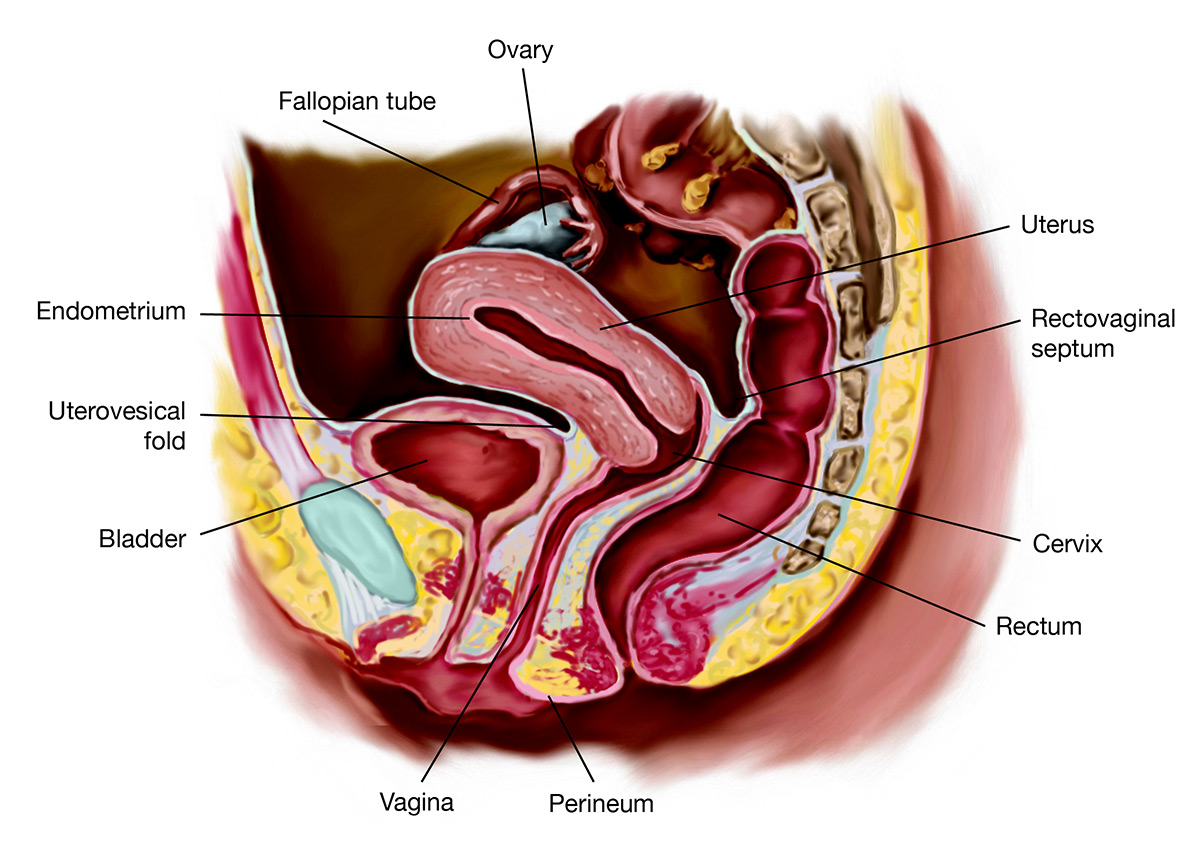
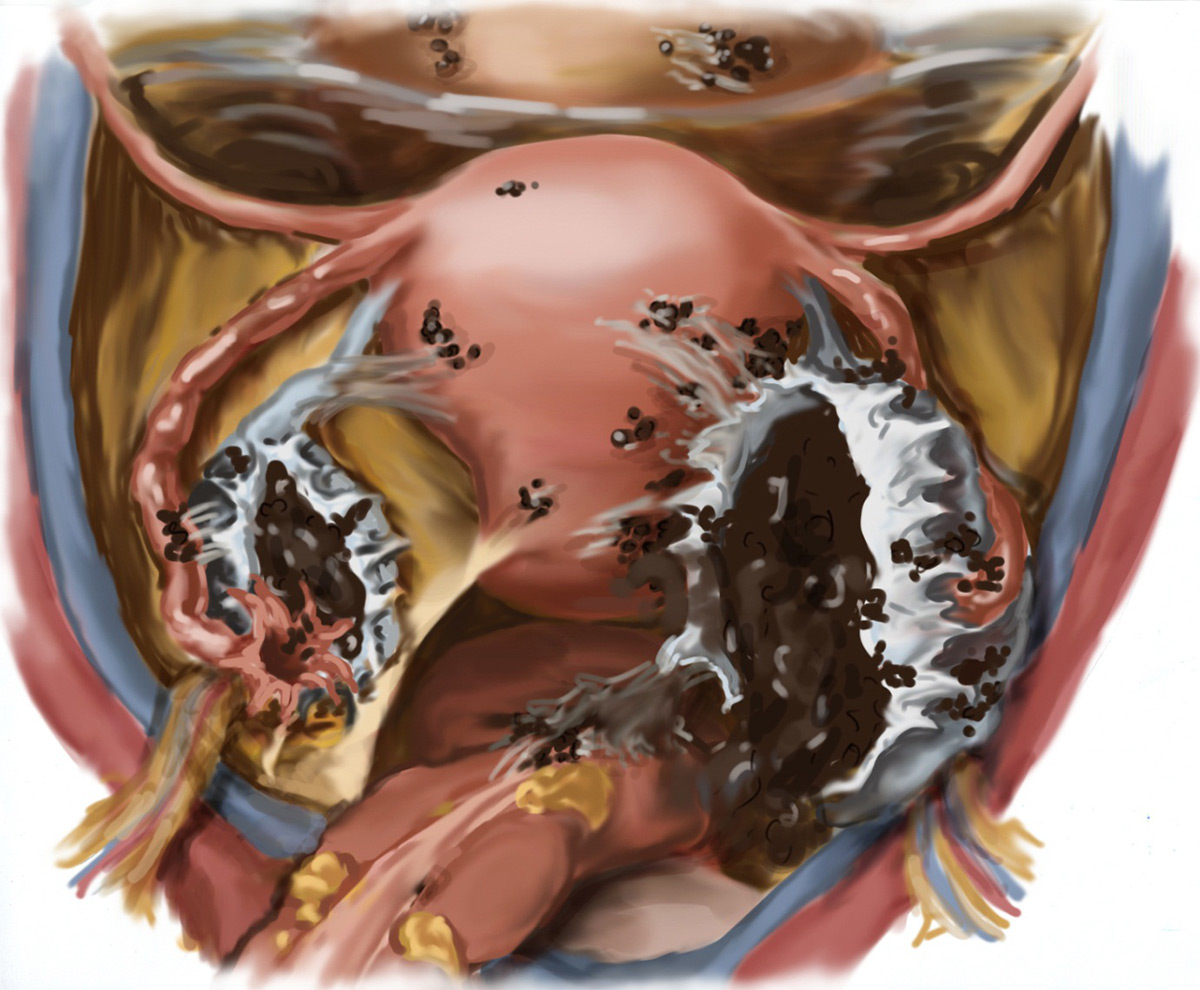
Symptomology
- Classic signs: severe dysmenorrhea, deep dyspareunia, chronic pelvic pain, Middleschmertz, cyclical or perimenstrual symptoms
- Typically develops on pelvic structures, ie, bladder, bowels, intestines, ovaries, and fallopian
- Less commonly found in distant regions, eg, diaphragm, lungs (inducing catamenial pneumothorax), and rarely, areas far outside abdominopelvic region
- Ovaries among most common of locations; gastrointestinal tract, urinary tract, soft tissues, and diaphragm follow
- Degree of disease present has no correlation with severity of pain or symptomatic impairment
Symptoms vary but typically reflect area of involvement and may include:
- Dysmenorrhea ( pain in menstrual bleeding)
- Heavy or irregular bleeding
- Cylical/noncylical pelvic pain
- Lower abdominal or back pain
- Bloating, nausea, and vomiting
- Dysuria (pain in urinating)
- Dyspareunia (pain while sexual intercourse)
Histiopathogenesis
- No single theory sufficiently explains pathogenesis
- Genetics, biomolecular aberrations in eutopic endometrium, dysfunctional immune response, anatomical distortions, and proinflammatory peritoneal environment may all ultimately be involved
- 5 key processes of development: adhesion, invasion, recruiting, angiogenesis, proliferation
Epidemiology
- More than 176 million women globally; 775,000 in Canada and 8.5 million in North America are affected
- Infertility among chief clinical findings
- No known prevention of Endometriosis
- Specific menstrual characteristics may be associated; decreased risk with late age at menarche and shorter menstrual cycles with longer duration of flow
- Family history cannot be undervalued; near 10-fold increased risk in women with first-degree relatives who have disease endometriosis
- No particular demographic, personality trait, or ethnic predilection
- Inverse BMI relationship
- No definitive association with nutrition, exercise, personality traits, or other lifestyle variables
Comorbidities
- Adhesions
- Risk of Adverse Pregnancy Outcome & Preterm Birth
- Up to 50% of those with endometriosis may suffer from infertility
- Distorted pelvic anatomy/impaired oocyte release or inhibit ovum pickup and transport
- Endocrine and anovulatory disorders
- Characterized as sexual dysfunction manifesting as pain in the reproductive organs before, during, or soon after sexual intercourse
- Though frequently depicted as psychogenic, dyspareunia (pain while intercourse) is actually often the result of organic, multidisciplinary cause and affects as many as 80% of endometriosis patients.
Diagnosis
- Clinical Diagnosis: Pelvic Examination & Pain Mapping, Medical History
- Imaging Studies, Ultrasound, MRI
- Surgical Diagnosis & Staging Surgical Intervention
Treatments
Surgical Interverntion
- Laparoscopy/Laparotomy
- Goals of conservative surgery include removal of disease, lysis of adhesions, symptom reduction and relief, reduced risk of recurrence, and restoration of organs to normal anatomic and physiologic condition
- Laparoscopic excision has been demonstrated to significantly improve deep dyspareunia as well as quality of sex life
Nonsurgical Therapies
- Medical treatment and combination therapy may help improve symptoms
- Gonadotropin-releasing hormone agonists (GnRH), oral contraceptives, Danazol®, Aromatase Inhibitors, and Progestins are mainstays
- Mirena® intrauterine device shown to be effective in reducing pain and may be considered alternative to hysterectomy in adenomyosis patients
- Alternative Therapies, for example herbal medicine, physical therapy, diet and nutrition, acupuncture, specific supplements and other complementary approaches may result in reduction of pain
Conclusion
- Endometriosis is a chronic, costly disease requiring long-term, multidisciplinary treatment
- Profound personal and economic impact underscores urgent need for continued research and improvement in diagnostic and treatment modalities
- Timely intervention and appropriate, multifactorial treatments may restore quality of life, preserve or improve fertility, and lead to long-term effective management in absence of permanent cure
For Consultation and individual Treatment Plan set up, please contact www.nicolasandasp.com
References
1. Eskenazi B, Warner ML. Epidemiology of endometriosis. Obstet Gynecol Clin North Am 1997;24:235–58.
2. Meuleman C, Vandenabeele B, Fieuws S, Spiessens C, Timmerman D, D'Hooghe T. High prevalence of endometriosis in infertile women with nor- mal ovulation and normospermic partners. Fertil Steril 2009;92:68–74.
3. Nnoaham KE, Hummelshoj L, Webster P, d'Hooghe T, de Cicco Nardone F, de Cicco Nardone C, et al. Impact of endometriosis on quality of life and work productivity: a multicenter study across ten countries. Fertil Steril 2011;96. 366–73.e8.
4. Berkley KJ, Rapkin AJ, Papka RE. The pains of endometriosis. Science 2005;
5. Simoens S, Dunselman G, Dirksen C, Hummelshoj L, Bokor A, Brandes I, et al. The burden of endometriosis: costs and quality of life of women with endometriosis and treated in referral centres. Hum Reprod 2012;27:
6. Crain DA, Janssen SJ, Edwards TM, Heindel J, Ho SM, Hunt P, et al. Female reproductive disorders: the roles of endocrine-disrupting compounds and developmental timing. Fertil Steril 2008;90:911–40.
7. Bulun SE. Endometriosis. N Engl J Med 2009;360:268–79.
8. Iwanoff N. Dusiges cystenhaltiges uterusfibromyom compliciert durch sar- com und carcinom. (Adenofibromyoma cysticum sarcomatodes carcino- matosum). Monatsch Geburtshilfe Gynakol 1898;7:295–300.
9. Levander G, Normann P. The pathogenesis of endometriosis; an experi- mental study. Acta Obstet Gynecol Scand 1955;34:366–98.
10. Merrill JA. Endometrial induction of endometriosis across Millipore filters. Am J Obstet Gynecol 1966;94:780–90.


